Potassium »
PDB 3ccm-3dke »
3ccm »
Potassium in PDB 3ccm: Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U
Protein crystallography data
The structure of Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U, PDB code: 3ccm
was solved by
G.Blaha,
G.Gurel,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 49.70 / 2.55 |
| Space group | C 2 2 21 |
| Cell size a, b, c (Å), α, β, γ (°) | 211.531, 298.177, 573.367, 90.00, 90.00, 90.00 |
| R / Rfree (%) | 20.1 / 24 |
Other elements in 3ccm:
The structure of Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U also contains other interesting chemical elements:
| Strontium | (Sr) | 108 atoms |
| Magnesium | (Mg) | 93 atoms |
| Cadmium | (Cd) | 5 atoms |
| Chlorine | (Cl) | 22 atoms |
| Sodium | (Na) | 75 atoms |
Potassium Binding Sites:
The binding sites of Potassium atom in the Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U
(pdb code 3ccm). This binding sites where shown within
5.0 Angstroms radius around Potassium atom.
In total 2 binding sites of Potassium where determined in the Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U, PDB code: 3ccm:
Jump to Potassium binding site number: 1; 2;
In total 2 binding sites of Potassium where determined in the Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U, PDB code: 3ccm:
Jump to Potassium binding site number: 1; 2;
Potassium binding site 1 out of 2 in 3ccm
Go back to
Potassium binding site 1 out
of 2 in the Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U
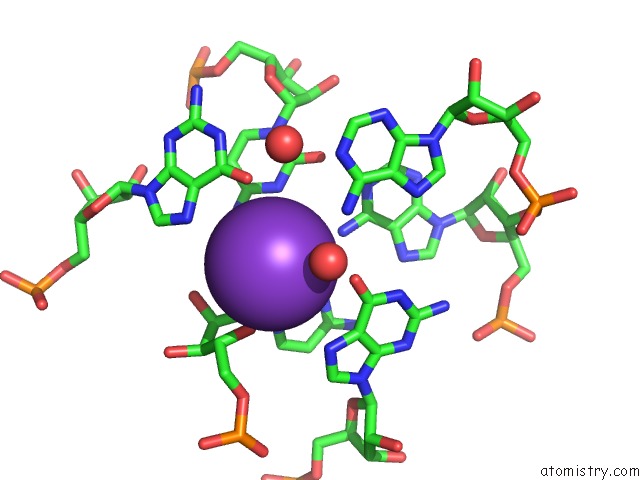
Mono view
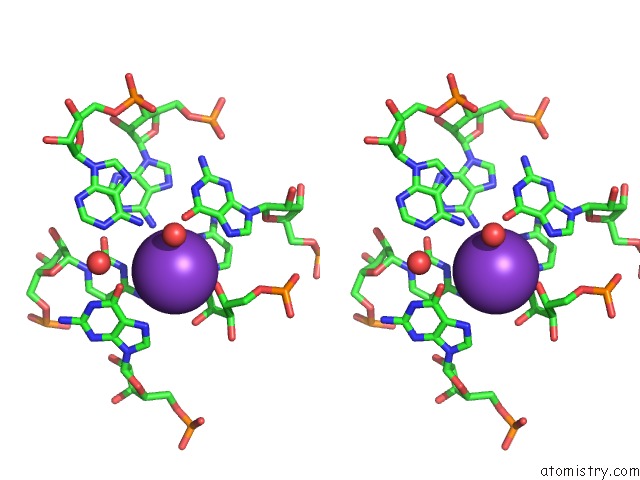
Stereo pair view

Mono view

Stereo pair view
A full contact list of Potassium with other atoms in the K binding
site number 1 of Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U within 5.0Å range:
|
Potassium binding site 2 out of 2 in 3ccm
Go back to
Potassium binding site 2 out
of 2 in the Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U
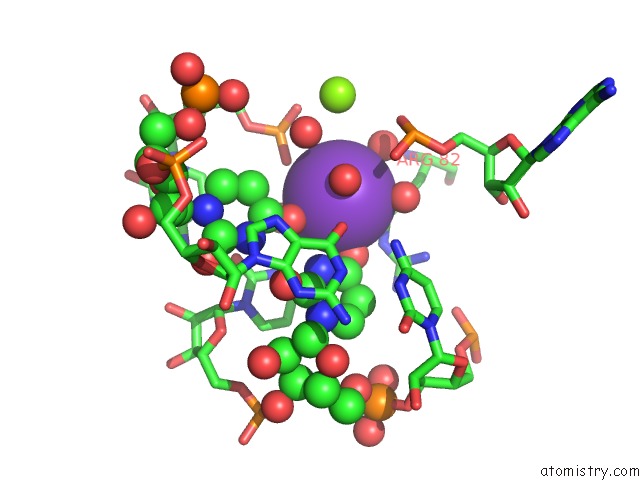
Mono view
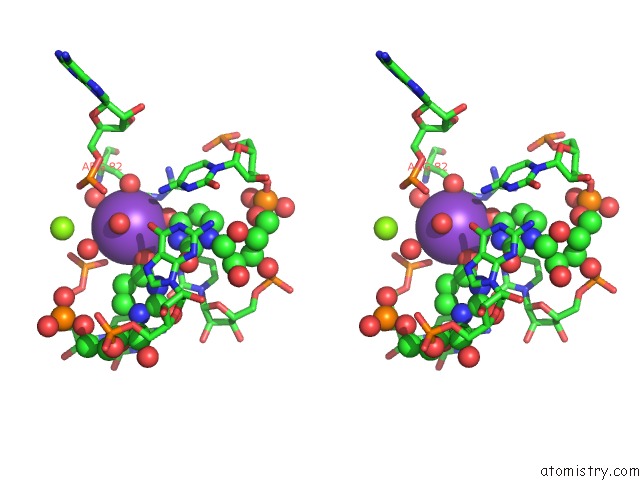
Stereo pair view

Mono view

Stereo pair view
A full contact list of Potassium with other atoms in the K binding
site number 2 of Structure of Anisomycin Resistant 50S Ribosomal Subunit: 23S Rrna Mutation G2611U within 5.0Å range:
|
Reference:
G.Blaha,
G.Gurel,
S.J.Schroeder,
P.B.Moore,
T.A.Steitz.
Mutations Outside the Anisomycin-Binding Site Can Make Ribosomes Drug-Resistant. J.Mol.Biol. V. 379 505 2008.
ISSN: ISSN 0022-2836
PubMed: 18455733
DOI: 10.1016/J.JMB.2008.03.075
Page generated: Mon Aug 12 07:57:07 2024
ISSN: ISSN 0022-2836
PubMed: 18455733
DOI: 10.1016/J.JMB.2008.03.075
Last articles
Zn in 9JYWZn in 9IR4
Zn in 9IR3
Zn in 9GMX
Zn in 9GMW
Zn in 9JEJ
Zn in 9ERF
Zn in 9ERE
Zn in 9EGV
Zn in 9EGW